
This and all of the behaviors represented by PV = nRT can be understood on the basis of a model called the Kinetic Molecular Theory. 2B: Gas Laws II (Worksheet) The fundamental relationship PV = nRT can be extended to understand the densities of gases under various conditions and to understand how non-reacting gases behave when mixed together.To understand how atoms combine to form compounds, you need to understand their basic composition and structure. The mass relationships between elements and compounds in chemical reactions ultimately relate back to the characteristics of the atoms of which they are composed. Atoms are the fundamental building blocks of all matter. 2A: Basic Atomic Structure (Worksheet) The atomic theory of matter is the great organizing principle of chemistry.This shows the relationship between a gas’s pressure (P), temperature (T), volume (V), and amount in moles (n). The observed behavior of gases, embodied in the empirical gas laws, leads to a series of equations that can be summarized by a single equation of state, called the ideal gas law equation. 1B: Gas Laws - Part 1 (Worksheet) Of the three principal states of matter (gas, liquid, solid), gases show behavior that is most easily connected to molecular motion.But regardless of the units used, we want to have some confidence that our measured and calculated results bear a close relationship to the “true” values. These units facilitate international communication by discouraging use of units peculiar to one culture or another (e.g., pounds, inches, degrees Fahrenheit). Since 1960, the metric system in use has been the Système International d'Unités, commonly called the SI units. 1A: Units, Measurement Uncertainty, and Significant Figures (Worksheet) All scientists the world over use metric units. 
Selenium-50 Selenium-of protonsof neutronsof electrons\) Sulfur-23 Sulfur-of protonsof neutronsof electrons Your periodic table and the information provided. Fill in the isotope names and any missing information on the chart. Assume all atoms are neutral.Ĭhromium-58 Chromium-of protonsof neutronsof electrons Nitrogen. For each of the following isotopes, write the number of protons, How can you tell isotopes of the same element apart?.What does the number next to isotopes signify?.Answer the questions based on the above reading. 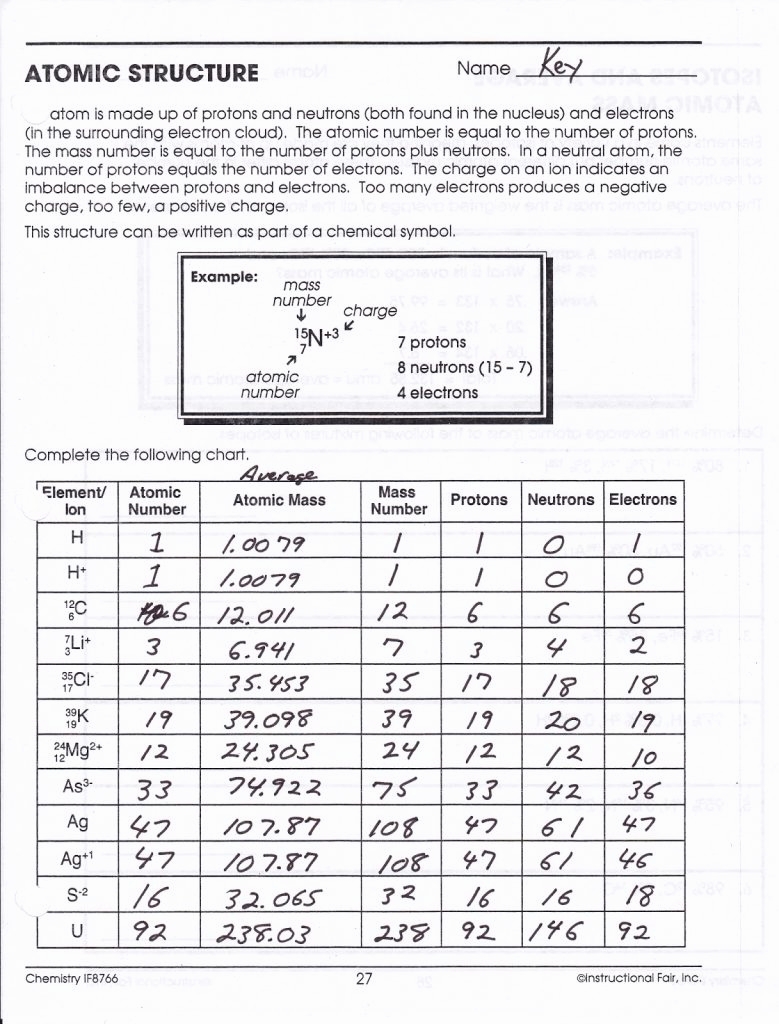
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
